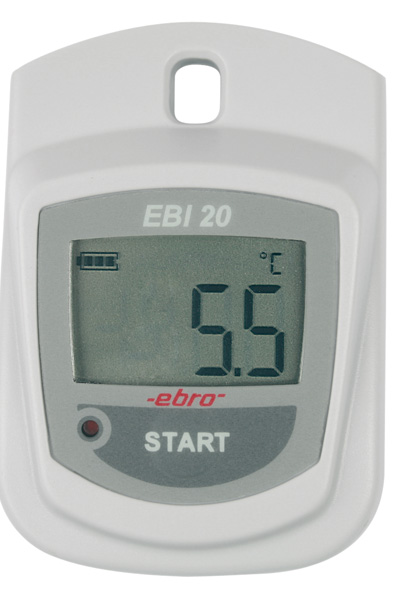
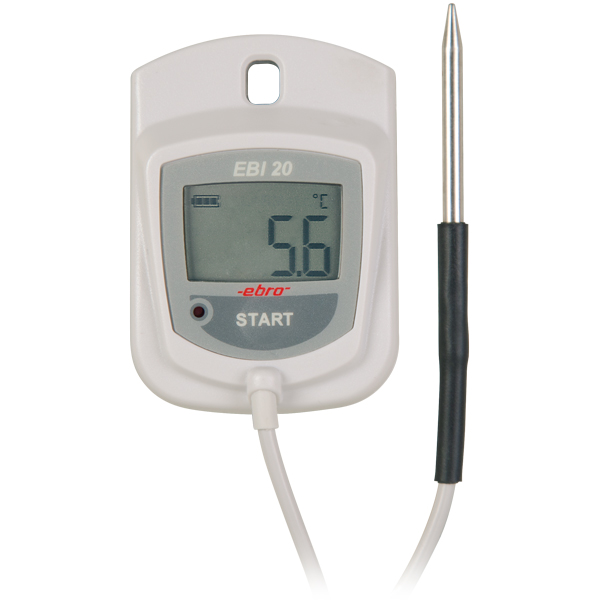
Pharmacie

The requirements of the EU GMP Guide and FDA publications pose major challenges for many manufacturers and suppliers
in the pharmaceutical sector. Our highly accurate and easy-to-use data loggers support you in the implementation of the
guidelines
and specifications regarding transport and logistics, warehousing and the validation and control of sterilization processes.
All ebro® software solutions enable user-friendly programming and reading of the data loggers and
are compliant with FDA
requirements regarding data integrity and 21 CFR Part 11.
Transports & Logistique Stockage Validation des processus de stérilisation Solutions logiciels
EBI 12-T231
Enregistreur de température pour la pasteurisation
Robust data logger for regular checks in canning factory.
EBI 11-T231
Enregistreur de données robuste pour le contrôle des échantillons
Robust temperature data logger for process validation of production processes.
EBI 12-T100-EX
Enregistreur de données de température polyvalent
Robust temperature data logger for regular independent measurements, even in hazardous areas.
EBI 310 TH
Enregistreur PDF
Surveillance de l’humidité relative dans les entrepôts et pendant le transport
EBI 11-T230
Enregistreur de données robuste pour le contrôle des échantillons
Robust temperature data logger for process validation of production processes.