Stockage

The current (c)GMP regulations such as "USP Chapter 1079 Good Storage and Distribution Practices for Drug Products"
set high standards for manufacturers and suppliers in the pharmaceutical sector. A risk-based approach must be used to ensure
that the temperature and relative humidity within the storage rooms are evenly distributed and that fluctuations can
be detected as quickly as possible.
Therefore, routine monitoring measurement points must be installed at clearly defined worst-case
positions. In order to identify areas that may endanger the quality of the stored products, a mapping of the storage area is
mandatory.
We are happy to support you with our GxP compliant mapping service.The ideal data loggers for routine monitoring
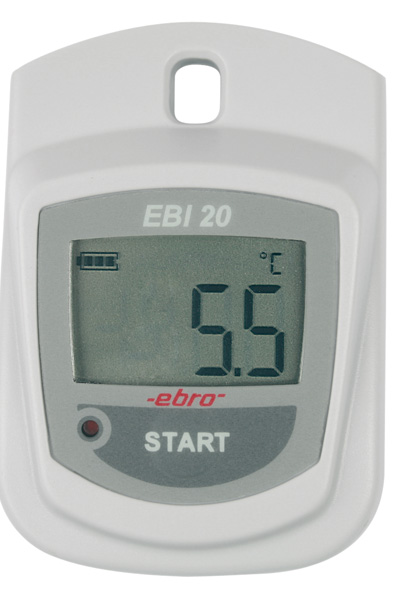
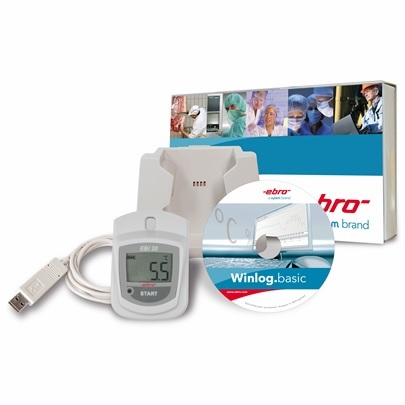
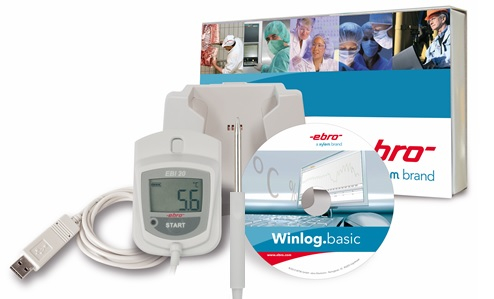
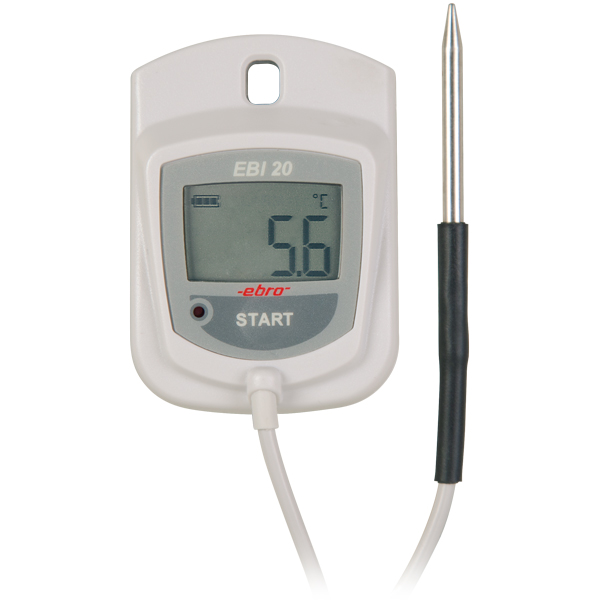
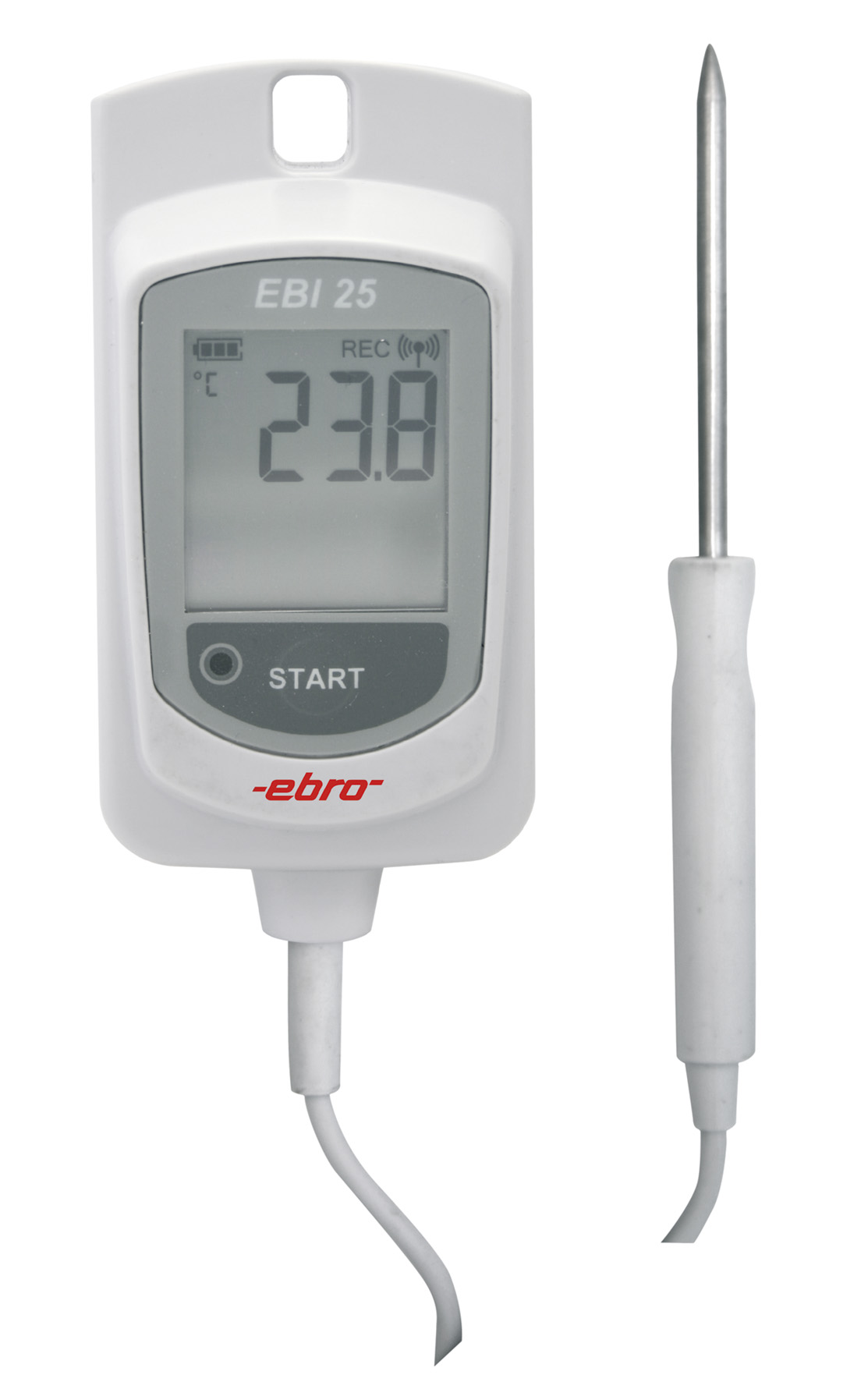
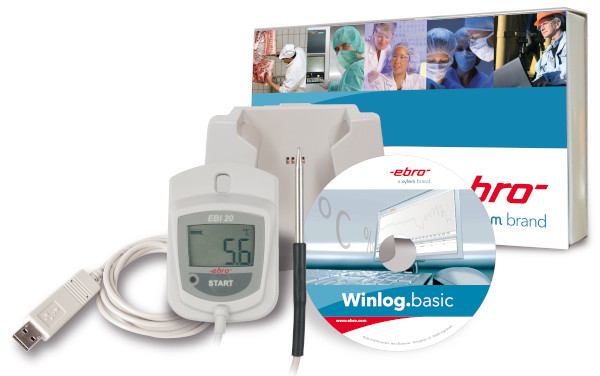
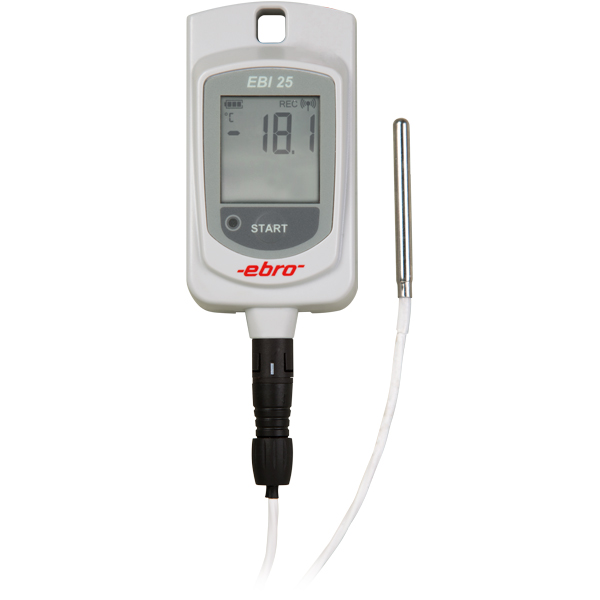
are the data loggers of the EBI-25 and EBI-20 family. Especially the EBI-25 wireless data loggers combine the very good
measurement accuracy and functionality of a data logger with the possibility to monitor up to 48 loggers via one interface.
With the Winlog.web software it is possible to visualize all measured values live on one screen. In addition, the alarm function
guarantees a quick response in case of temperature or humidity deviations. The IQ and OQ can either be performed by
our qualified staff or we can provide you with the necessary documents.
set high standards for manufacturers and suppliers in the pharmaceutical sector. A risk-based approach must be used to ensure
that the temperature and relative humidity within the storage rooms are evenly distributed and that fluctuations can
be detected as quickly as possible.
Therefore, routine monitoring measurement points must be installed at clearly defined worst-case
positions. In order to identify areas that may endanger the quality of the stored products, a mapping of the storage area is
mandatory.
We are happy to support you with our GxP compliant mapping service.The ideal data loggers for routine monitoring
are the data loggers of the EBI-25 and EBI-20 family. Especially the EBI-25 wireless data loggers combine the very good
measurement accuracy and functionality of a data logger with the possibility to monitor up to 48 loggers via one interface.
With the Winlog.web software it is possible to visualize all measured values live on one screen. In addition, the alarm function
guarantees a quick response in case of temperature or humidity deviations. The IQ and OQ can either be performed by
our qualified staff or we can provide you with the necessary documents.