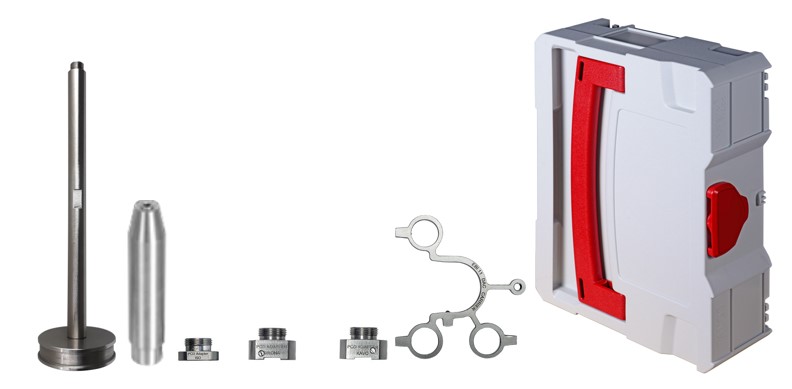
SL 3302 Complete Validation Set
for DAC Universal, benchtop autoclaves as well as washer disinfectors
Very flexible data logger system to perform validations for DAC Universal MK IV according to ISO 15883. This reliable system consists of user-friendly mini data loggers that can be placed directly in the processes and an evaluation software package that has been certified by the TÜV.
The set contains:
- 2 x EBI 11-T235 Mini Temperature data logger, Needle length = 25 mm
- 2 x EBI 11-T236 Mini Temperature data logger, Needle length = 80 mm
- 1 x EBI 11-P111 Mini Pressure data logger
- EBI IF 300, 4-port Interface
- Winlog.validation software
- 1 x Carrying case SYSTAINER
-
The EBI 11-T235 validation logger was specially developed for the validation of reprocessing of medical devices in the tertiary sector. It is adapted to the validation concept for the Sirona DAC Universal and Melag Careclave units.
In addition to being used in doctors surgeries, the logger can also be used to measure the temperature in the endoscope dummy. The needle length is precisely matched to the AL 126 adapter.
Benefits:
- simple and repeatable placement
- wireless measurement
- small size
- high accuracy
Applications:- validation at doctors surgeries
- process validation with DAC Universal
- process validation with Careclave
- process validation with endoscope-dummy,
Spypach Process validation - Process validation
DatasheetProduct number: 1340-6270 -
Miniature pressure data logger for measuring the temperature-compensated absolute pressure during validation with small sterilizers and cassette autoclaves. The data logger can also be used in RDGs as well as in the Sirona DAC Universal and Melag Careclave. With its Luer lock connection, it can be used universally.
Benefits:
- small size
- large measuring range
- wireless measurement
- connectable to Luerlock
- high accuracy
Applications:- process validation with DAC Universal
- process validation with Careclave
- process validation with endoscope-dummy, Spypach
- F0 Value calculation
DatasheetProduct number: 1340-6296 -
The EBI 11-T236 validation logger was specially developed for the validation of reprocessing of medical devices in the tertiary sector. It is adapted to the validation concept for the Sirona DAC Universal and Melag Careclave units.
Benefits:
- simple and repeatable placement
- wireless measurement
- small size
- high accuracy
Applications:- validation at doctors surgeries
- process validation with DAC Universal
- process validation with Careclave
Product number: 1340-6271 -
With the Adapter set is an independent process qualification of a DAC Universal MK III and MK IV incombination with ebro EBI 11 data loggers possible. No modification on the customers lid necessary.The set contains:• 1 x Logger mount
• 1 x PCD test body incl. adapter for ISO, Sirona and KaVo
• Carrying case „SYSTAINER“Product number: 1248-3305 -
For transport of systainers Fixed seat of the systainers on the trolleyProduct number: 1248-0127
NEW: Information package "Professional data loggers"
Please fill out the form below and the download will start instantly.